As endotoxins are exposed within the surface of microorganisms, the innate immune program has progressed to recognise them like a menace and also to react accordingly for their existence. Endotoxins are pyrogens, provoking a powerful innate immune reaction. When Gram-detrimental micro organism are killed through the immune method, fragments of their membrane containing endotoxins are launched during the blood stream and will induce fever and diarrhoea.
The specific mechanism of the phenomenon, termed endotoxin tolerance, is unfamiliar". Also, some scientific tests have proven which the endotoxin with the Legionnaires' bacillus has a special spectrum of toxicity than the greater widespread gram-detrimental organisms. Specifically the Legionnaires' endotoxin was not extremely pyrogenic (via the rabbit test), but was quite Energetic during the LAL - one,000 fold difference between the two tests. In this case, the rabbit test is insufficient for figuring out the potency of the toxins existing.
It leverages the blood cells of horseshoe crabs, which coagulate within the presence of endotoxins, offering a sensitive implies to detect their presence. Even with its efficacy, ethical and sustainability problems about horseshoe crab populations have prompted the event of different methods.
Among the list of most vital virulence things of endotoxins is definitely the inflammatory reaction they may cause in human hosts.
Great apply would come with control of the microbiological and endotoxin levels of contamination inside the likely sources pointed out higher than.
Endotoxins serve as an early diagnostic biomarker to serologically recognize Gram-unfavorable-distinct bacterial bacterial infections. Timely identification is indispensable for early illness therapy.
Developments in nanotechnology have released progressive methods to endotoxin neutralization. Nanoparticles, created to mimic the framework of host cell membranes, can competitively bind to endotoxins, therefore blocking them from interacting with immune cells.
Other significantly less frequently acceptable methods contain ethylene oxide treatment method and irradiation. It's been shown that reductions of close to 80% from the pyrogenicity here of E. coli endotoxin in dialyzers have happened after publicity to ethylene oxide.
In contrast, the Monocyte Activation Test (MAT) offers another solution by mimicking the human immune reaction to endotoxins. This assay takes advantage of human blood cells to detect the inflammatory reaction activated by endotoxins, giving insights into their Organic exercise rather than just their existence.
Actually, endotoxins can only be passively released. This normally takes place either by means of Dying, mechanical harm and lysis of microorganisms but additionally throughout bacterial progress and division.
Poster: Fluorescence-dependent bacterial endotoxin testing – Superior sensitivity detection with a versatile and streamlined workflow
Endotoxins create a barrier that may be permeable only to hydrophilic molecules with small molecular bodyweight, generating Gram-damaging germs resistant to lots of antimicrobial compounds.3
When purifying plasmids and transfecting endotoxin-delicate cell lines, to maintain cell well being and transfection performance
We’ve got you included, whether or not you’re just getting started with endotoxin Investigation or willing to dive into here large-throughput applications plus more.
 Hallie Eisenberg Then & Now!
Hallie Eisenberg Then & Now! Michael J. Fox Then & Now!
Michael J. Fox Then & Now! Michael C. Maronna Then & Now!
Michael C. Maronna Then & Now! Danny Pintauro Then & Now!
Danny Pintauro Then & Now!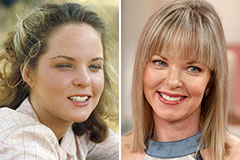 Melissa Sue Anderson Then & Now!
Melissa Sue Anderson Then & Now!